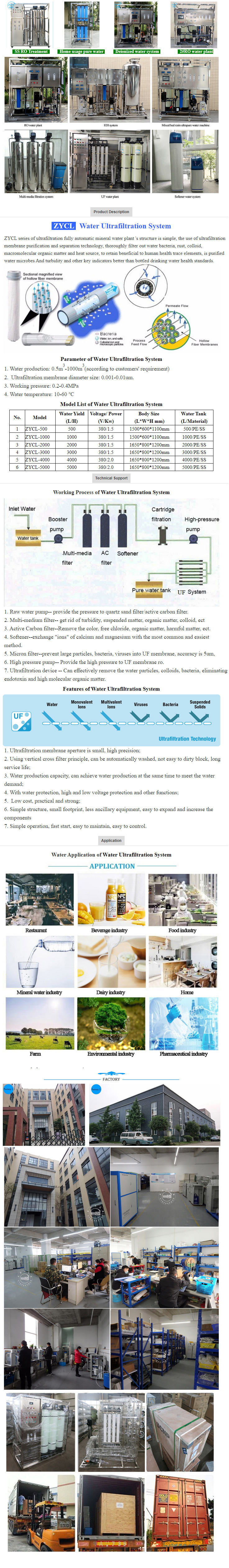
Pharmaceutical companies GMP-2
Product description
As China's medical standards, considerable development of biological science, people of the pharmaceutical industry are increasingly high requirements. Many pharmaceutical companies have recognized manufacturers: To ensure the quality of pharmaceutical products to prevent contamination of the product production environment, production areas must meet the standards prescribed by the state environmental parameters. Pharmaceutical industrial clean rooms and clean area requirement particles and microorganisms in the main control object from the environment, but also respond to temperature, humidity, fresh air quantity, pressure, illumination, noise and other parameters made??, make the necessary provisions. Our company strictly in accordance with: international standard "ISO / DIS 14644", and clean plant design specifications "GB50073-2001", the pharmaceutical industry clean plant design specifications "GMP-7", pharmaceutical production and quality management practices "GMP-98", ventilation and air-conditioning engineering construction and acceptance "GB50243-2002" and other national standards. Design and construction of a large number of pharmaceutical companies GMP standard workshop, and passed the relevant state departments of acceptance.
Previous
Drug filling workshop
Next
Pharmaceutical plant
Related products
Chaozhou Saite Purifying Apparatus Co., Ltd.,
Hotline:
Add:
Liyangli Industrial Area, Dongfeng, Chaozhou City, Guangdong Province
Copyright © 2021 Chaozhou Saite Purifying Apparatus Co., Ltd., 粤ICP备2023091047号







 0768-6581475
0768-6581475

 Messages
Messages 